in this article
authors
How close are we to understanding the brain? “We don’t even understand the brain of a worm,” said Christof Koch, Ph.D., Meritorious Investigator at Allen Institute.
The lab roundworm, more technically known as Caenorhabditis elegans, houses 302 neurons and 7,000 connections between those neurons in its microscopic body. Researchers have painstakingly mapped and described all those connections in recent years. And we still don’t fully understand how they all work synergistically to give rise to the worm’s behaviors.
We humans have approximately 86 billion neurons in our brains, woven together by an estimated 100 trillion connections, or synapses. It’s a daunting task to understand the details of how those cells work, let alone how they come together to make up our sensory systems, our behavior, our consciousness.
We asked Koch and his neuroscience colleagues to reflect on how much we still don’t know about the brain, and how these researchers are trying to solve those mysteries.
What is the brain made of?
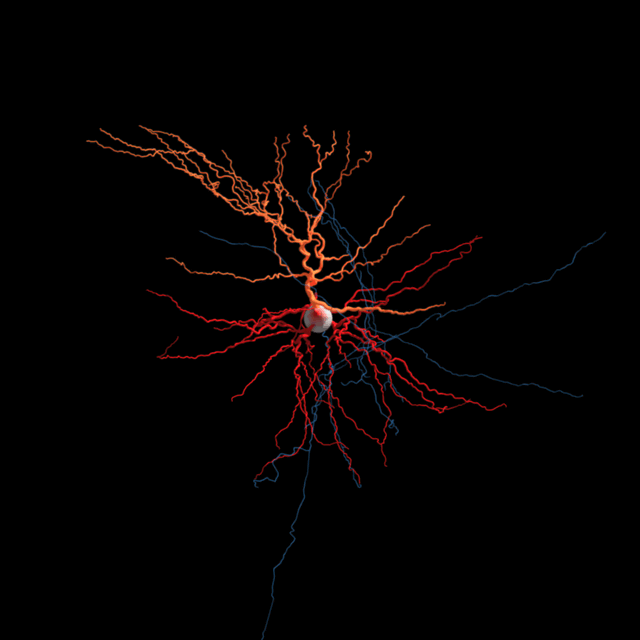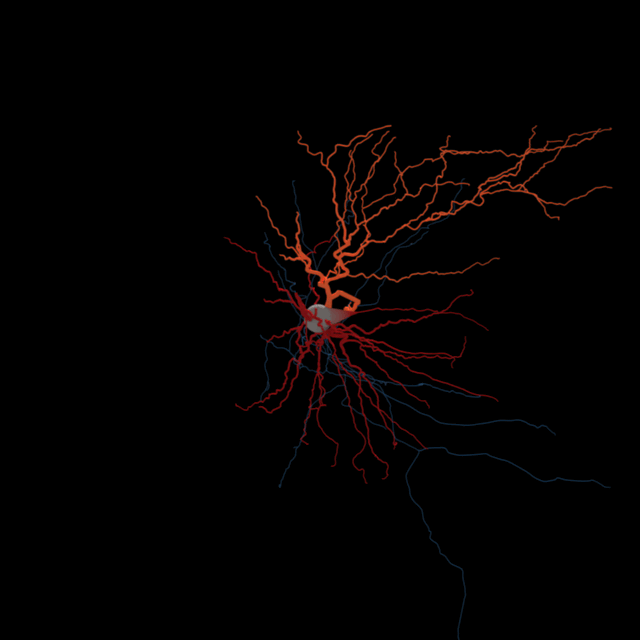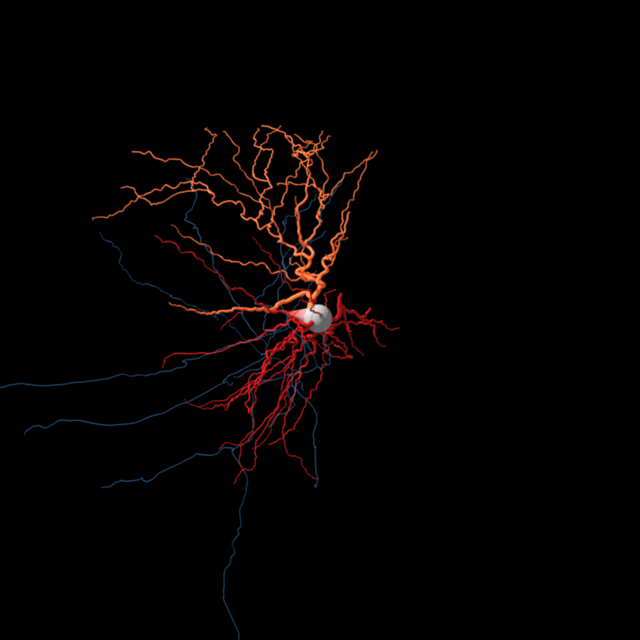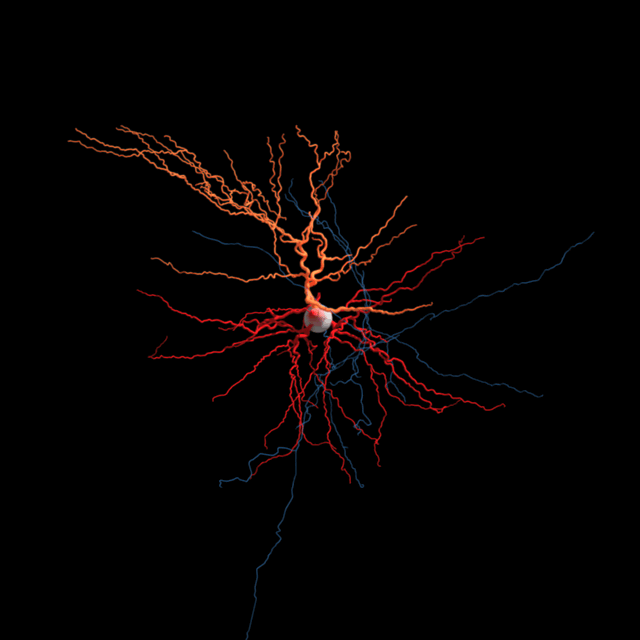
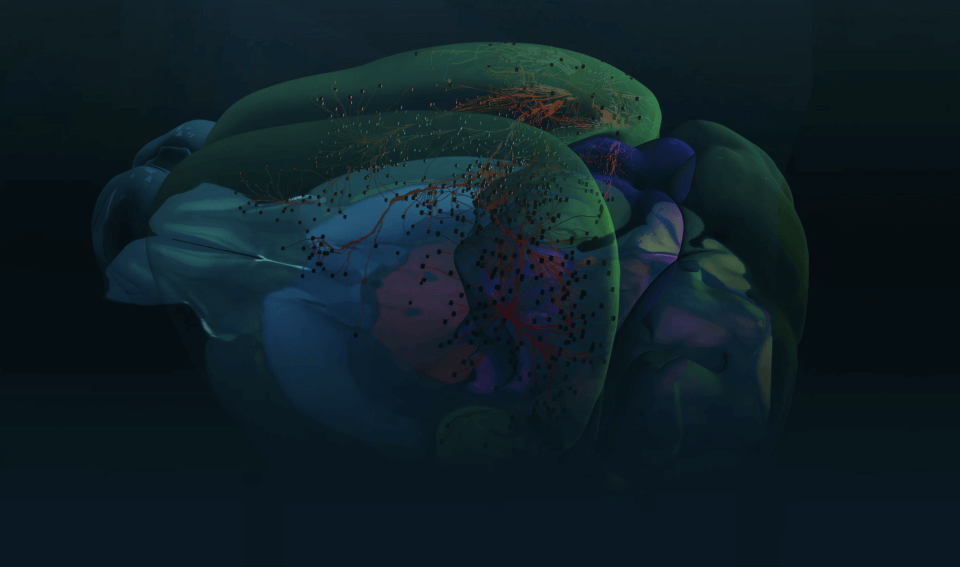
The brain consists most obviously of gray matter and white matter, brain tissue and its interconnections or bundles of axons. Look more closely at the former and one can distinguish neurons and glia (the other kind of brain cell). But we’re far from understanding all the types of neurons and other brain cells at the level of what they do.
“How can we understand the entire thing if we don’t understand how many different components there are?” Koch asked.
He and his colleagues sometimes refer to this as discovering the “periodic table of brain cell types.” Chemists have an organized table that describes the 118 known chemical elements — the building blocks of matter — but neuroscientists are lacking such a well-defined categorization of the brain’s building blocks.
It’s human nature, or at least many scientists’ nature, to understand something by categorizing it. When Koch was a kid, the first thing he’d do with a new box of Legos was to sort them into types, he said: “The one-by-one, the one-by-two, the two-by-four, etc.”
Sorting neurons is not as simple. Allen Institute for Brain Science researchers are using several characteristics to define a brain cell type. Different teams at the Institute are sorting cells based on the genes they switch on and off, their detailed shapes, the regions of the brain they connect to, and their unique electrical behavior. Then comes the hard task of putting all that information together to define brain cell types based on all of these attributes.
How does the brain change in disease?
A big part of understanding the brain’s parts list is so researchers can better understand which cells in the brain might underlie neurological and psychiatric diseases. Many neuropsychiatric disorders do not affect the entire brain uniformly, but rather start in or are driven by specific classes of neurons or other brain cells.
“Right now, we don’t understand which cell types are vulnerable in these diseases,” said Boaz Levi, Ph.D., a neuroscientist at the Allen Institute for Brain Science.
If researchers compile the full list of brain cell types, they could then see which types of cells die, grow out of control or otherwise change their course in diseases of the brain. Researchers could then build better tools to study those disease-triggering cells — and possibly therapies that target a single cell type at the heart of disease.
As part of the Allen Institute’s work studying different human brain cell types, Levi and his colleagues develop molecular tools to isolate and track those specific cells. Those tools could potentially be engineered to deliver specific gene therapies or other treatments directly to a certain cell type. The Allen Institute researchers are now collaborating with a team at Seattle Children’s Research Institute to test whether one of these tools could be used to treat Dravet syndrome, an uncommon but severe form of early-childhood epilepsy that is usually caused by a mutation in a single gene and which affects a specific class of neurons.
How do neurons talk to each other?
Biology textbooks tell us that the brain communicates via synapses, specialized connections between two different neurons.
“We believe this is true for many neuron types in the brain,” said Jack Waters, Ph.D., a neuroscientist at the Allen Institute for Brain Science.
The majority of neurons use one of two common signaling molecules known as neurotransmitters, GABA or glutamate, that are known to pass through specialized synapses. But there are many other types of signaling molecules present in the brain, and it’s not clear how those molecules get their message across.
Take, for example, the molecules that most neurological or psychiatric drugs act on.
“If you were to scroll through all the drugs that people have heard of, most of them are not acting on glutamate or GABA,” Waters said. “With drugs like opioids or antidepressants, we actually don’t understand the mechanisms of the underlying molecules those drugs are interacting with.”
It’s a difficult question to answer because it’s so broad, Waters said. But data gathered through a collaborative project known as the IARPA MICrONS project could help. That work, which is partly conducted at the Allen Institute, is creating the largest ever roadmap of connections in the mammalian brain, mapping a piece of the mouse visual cortex the size of a grain of sand that contains about a billion synapses. Once that’s complete, researchers can start to piece together the puzzle of which molecules go with which synapses, Waters said.
How does the brain compute?
If understanding the brain’s makeup is a challenge, figuring out how those billions of components come together to enable all the brain’s complex behavior is even more difficult. The Allen Brain Observatory team aims to capture a small part of that complexity: how a mammal’s brain represents and processes visual information.
Neuroscientists have been studying the visual part of the mammalian brain for decades, but until very recently technology only allowed them to capture information from a handful of neurons at a time. It’s like if you tried to watch a movie but could only see 1000 pixels out of several million on the screen, Koch said.
“Imagine you have to infer who loves whom, who backstabs whom, what’s going on from just those few pixels,” he said. “That’s the situation you have had in neuroscience until recently. You record from a handful of neurons and try to infer some common principles.”
Researchers on the Observatory team are now looking at tens of thousands of neurons as they fire in real time. As for those principles of computation? So far, there doesn’t seem to be a simple answer, Koch said.
What will it mean to understand our brains?
Rebecca Hodge, Ph.D., a scientist at the Allen Institute for Brain Science, holds a frozen slice of a postmortem human brain donated to research. A new effort led by the Allen Institute aims to map the entire human brain at single-cell resolution. Photo by Erik Dinnel / Allen Institute
When we think about understanding something, we often think about being able to explain it in a relatively simple way. In science, researchers in other fields look to physics as a model of understanding, said Koch, who is himself a former physicist. The physical world lends itself to abstractions that can be boiled down to (relatively) simple equations.
But what if biology doesn’t? The more Koch and others at the Allen Institute study the brain at large-scale, looking at many or most cells in the brain rather than just a few, the more they realize that even the parts of neuroscience they thought the field had nailed down are more complicated than anyone had realized.
“There may not be any simple path to understanding complex systems shaped by natural selection,” Koch said. “Evolution doesn’t care about elegance. The brain doesn’t care if you understand it.”
So how can we get to an understanding of the brain that will help feed medical research and satisfy our curiosity about this organ that makes us uniquely who we are? It might take more computational power, Koch said. Computer models can help, but we may need a lot of them to explain each little piece of the puzzle. Or it might just mean embracing the power of big data.
The good news is that technology has advanced to the point where we can gather and store that data in larger and larger amounts. And in recent years, there’s been a growing interest in and funding for neuroscience, thanks in part to the 2013 BRAIN Initiative through the National Institutes of Health.
“This is a golden era of brain science,” Koch said. “There are so many more resources and institutes — like ours — where we can pursue questions that were really unthinkable 20 years ago.”
Citations
about the allen institute
The Allen Institute is an independent, 501(c)(3) nonprofit research organization founded by philanthropist and visionary, the late Paul G. Allen. The Allen Institute is dedicated to answering some of the biggest questions in bioscience and accelerating research worldwide. The Institute is a recognized leader in large-scale research with a commitment to an open science model. For more information, visit alleninstitute.org.