Trygve Bakken
Investigator, Assistant
Trygve Bakken maps the vast diversity of cell types in the human brain and compares them to other species to understand how our brains develop, evolve, and change in disease. By pairing large-scale single-cell genomics with genetic tools for precise circuit access, his work lays the groundwork for new approaches to study and treat brain disorders. Dr. Bakken joined the Allen Institute in 2013 after earning his M.D. and Ph.D. in Neuroscience at the University of California, San Diego, where he investigated the genetic basis of human brain morphology and developmental principles of neural circuitry. He also holds an M.Sc. in Philosophy and History of Science from the London School of Economics and a B.A. in Physics and Philosophy from Yale University.
research focus
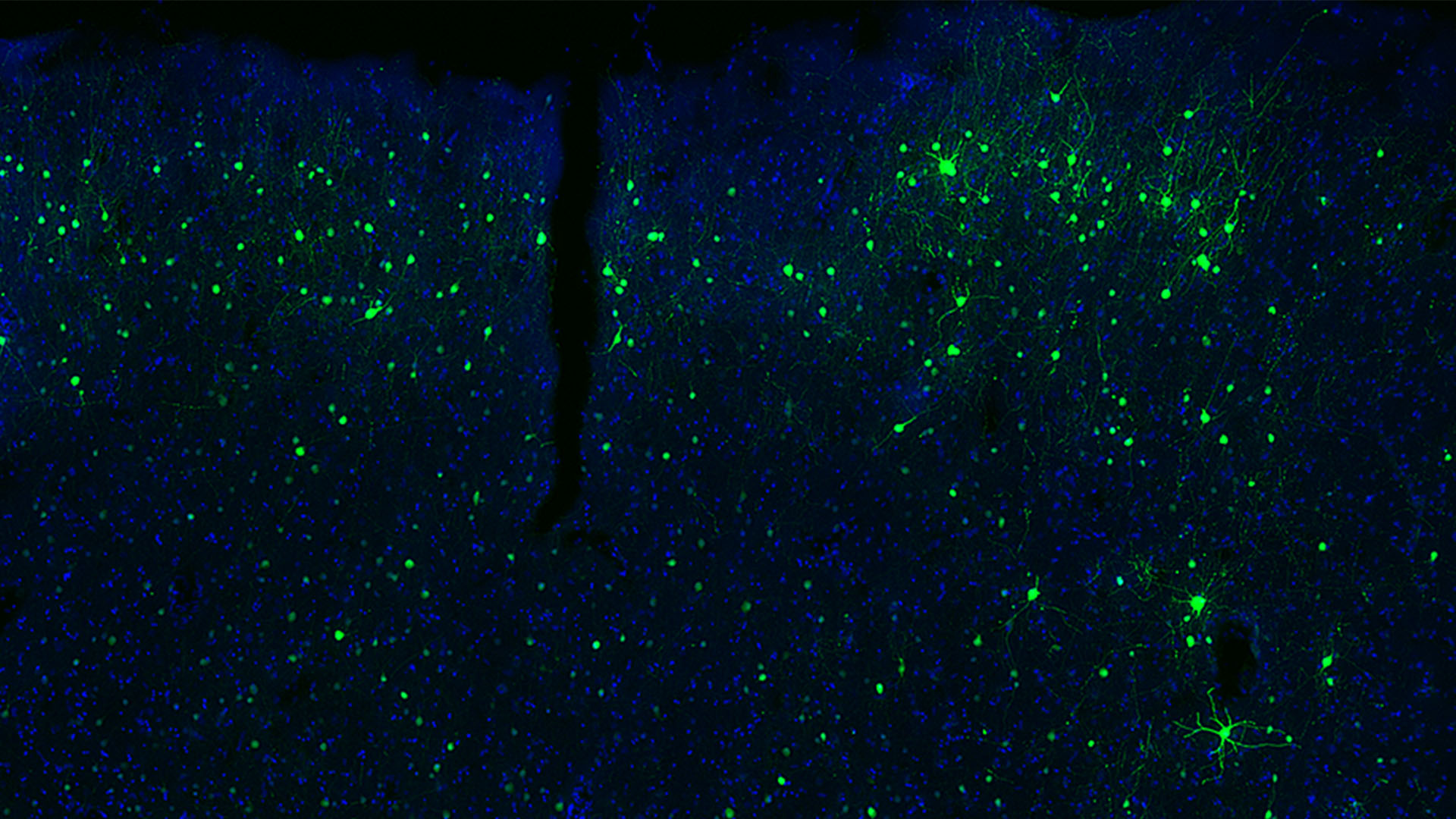
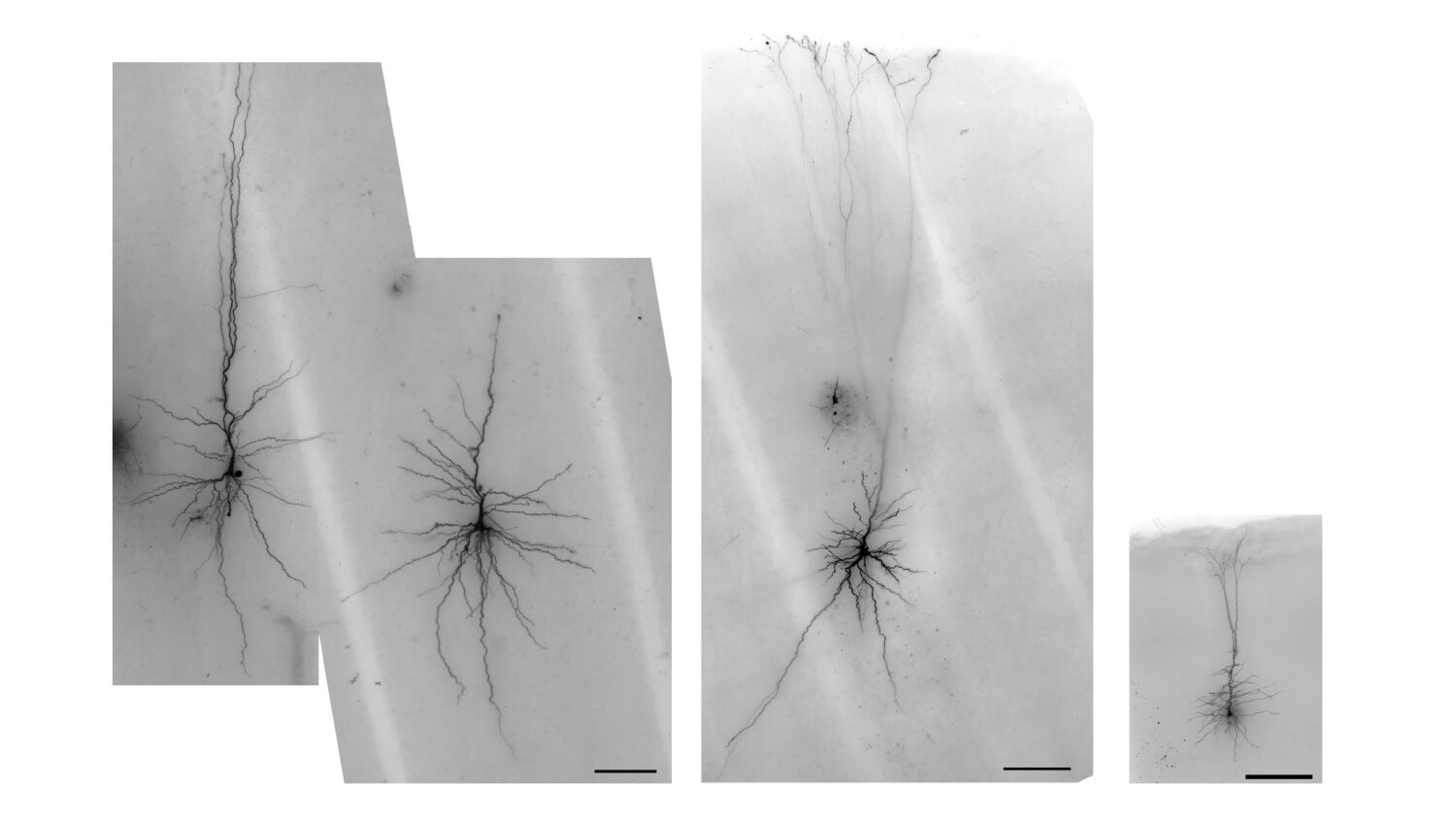
Dr. Bakken leads cross-species studies of neural cell type diversity using high-throughput, multiomic profiling of single nuclei from the human brain and other mammals. His research defines conserved and human-specialized molecular phenotypes of mammalian brain cell types. His team develops machine learning models to predict in vivo activity of cell type-specific enhancers, enabling the systematic design of viral tools for precise access to targeted neuronal and non-neuronal populations. These enhancer-AAV tools, validated across rodents and non-human primates and distributed via the Genetic Tools Atlas, provide standardized resources for probing neural circuits and informing therapeutic strategies.In earlier studies, Dr. Bakken generated transcriptional atlases of primate cortical development, revealing conserved and uniquely human developmental trajectories and implicating specific cell populations in psychiatric risk for autism spectrum disorder and schizophrenia. He has also linked variation in cell type abundance and gene expression to age, sex, genetic background, and evolutionary history, and demonstrated how ancient human population movements have shaped cortical architecture. Ongoing work extends these approaches across additional human donors and species to connect molecular, developmental, and functional perspectives on brain organization, evolution, and disease.