in this article
authors
Researchers at the Allen Institute for Brain Science, UW Medicine, and Kaiser Permanente Washington Health Research Institute have created the most detailed picture yet of how Alzheimer’s disease (AD) progresses at the cellular level.
Using advanced single-cell genomic technologies and novel machine learning models, the scientists mapped out a timeline of the disease’s cellular and molecular changes. In the process, they identified a specific type of inhibitory neuron (the somatostatin-expressing inhibitory neuron) as one of the earliest cell types that is lost in AD—a surprising discovery that could highlight potential targets for future therapies.
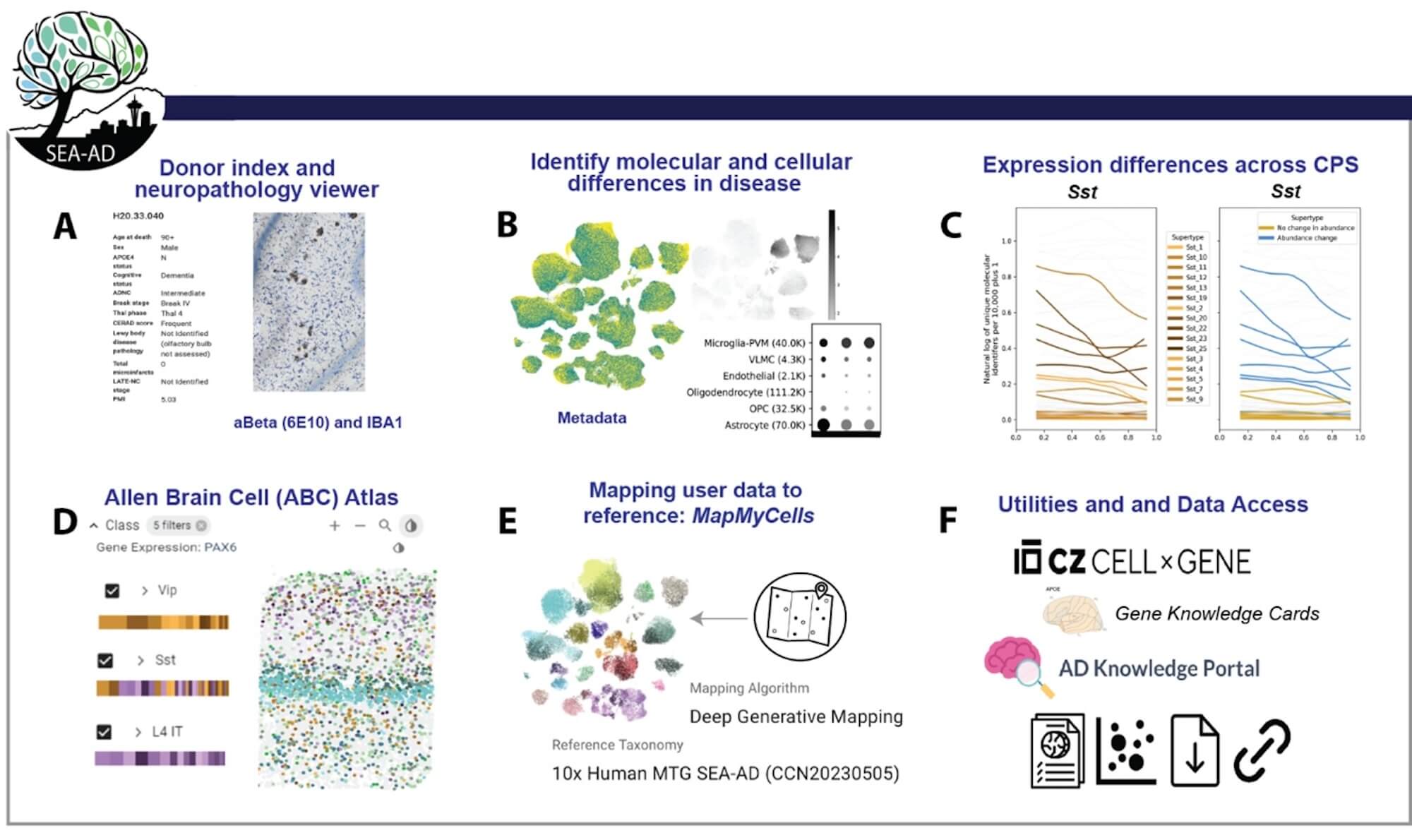
The study, published today in Nature Neuroscience, analyzed over 3.4 million cells from 84 brains donated by Alzheimer’s disease research participants. By making this massive dataset available through the Seattle Alzheimer’s Disease Brain Cell Atlas (SEA-AD), a consortium supported through the National Institute on Aging (NIA), the scientists hope to accelerate global research.
“The takeaway is that this atlas describes AD progression at unprecedented cellular resolution, and identifies many new cellular and molecular targets for the field to explore,” said Kyle Travaglini, Ph.D., a lead author and scientist at the Allen Institute.
Creating a pathology clock
The study zoomed in on a region of the cortex called the middle temporal gyrus (MTG), which is involved in language, memory, and higher-order visual processing. The MTG is also a critical transition zone where preclinical Alzheimer’s pathology, like the buildup of toxic protein fragments, transitions to more advanced neurodegeneration linked to dementia.
The new knowledge provided by this study may help scientists and drug developers around the world develop diagnostics and treatments targeted to specific stages of Alzheimer’s and other dementias.
John Ngai, Ph.D., Director of The BRAIN Initiative®
To explore this progression, the researchers harnessed single-cell and spatial genomics technologies developed with funding by the National Institutes of Health’s (NIH’s) BRAIN Initiative Cell Census Network (BICCN). They used these tools to map the active genes, DNA structure, and precise location of individual cells in the MTG region in AD brain samples. They then compared that data to a massive cell-type normal brain reference map generated earlier by the Allen Institute team and their BICCN collaborators.
“This research demonstrates how powerful new technologies provided by the NIH’s BRAIN Initiative are changing the way we understand diseases like Alzheimer’s. With these tools, scientists were able to detect the earliest cellular changes to the brain to create a more complete picture of what happens over the entire course of the disease,” said John Ngai, Ph.D., director of The BRAIN Initiative®. “The new knowledge provided by this study may help scientists and drug developers around the world develop diagnostics and treatments targeted to specific stages of Alzheimer’s and other dementias.”
The 84 research participant brain donors were from UW Medicine Alzheimer’s Disease Research Center (ADRC) and the Kaiser Permanente Washington Adult Changes in Thought (ACT). Donors ranged from those with advanced Alzheimer’s dementia to unimpaired subjects without evidence of AD. “By studying research subjects across the spectrum of AD, including those in the earliest stages of disease, we hope to identify vulnerable cells early in the disease process, long before a person develops symptoms,” says C. Dirk Keene, Professor and Nancy and Buster Alvord endowed chair in Neuropathology at UW Medicine. The team also analyzed detailed neuropathology data to model disease progression along the continuous spectrum of pathology characterized by this cohort.
Using custom machine learning tools to align the datasets, they constructed an unprecedented high-resolution view of how AD impacts different cell populations over time.
“You could say that we created a pathology clock that tells not only what changes are happening in this cortical region, but when,” said Mariano Gabitto, Ph.D., a lead author and assistant investigator at the Allen Institute. “We now have a framework to arrange the sequence of events as Alzheimer’s pathology increases over time.”
Early vs. late impacts
The researchers identified two distinct phases of AD: a slow, early buildup of abnormal cellular changes that occurs before any memory or cognitive impairments appear, followed by a later rapid increase that coincided with cognitive decline.
In the early phase, they found inflammatory changes in the brain’s immune cells (microglia) and support cells (astrocytes), as expected from studies of genetic risk for AD that implicate those non-neuronal cells. They also identified specific types of neurons that are lost very early in disease: a specific type of inhibitory interneuron that helps dampen neural activity.
The loss of these “SST neurons” (somatostatin-expressing inhibitory neurons) was a surprise. Most of the field has focused on microglia, and a loss of excitatory neurons that make long-range connections across the cortex and other brain regions, said Ed Lein, Ph.D., senior investigator at the Allen Institute for Brain Science and lead investigator on the SEA-AD team. Instead, we find it is specific types of inhibitory neurons that are the earliest neuronal casualties in this part of the brain.
They also observed a loss of cortical oligodendrocytes, cells that insulate nerve fibers and help speed up communication in the brain. That decline was followed by the activation of a repair program to restore this insulation.
As the disease progresses, the later phase involves much more extensive loss of neurons, including specific types of excitatory and inhibitory neurons. Strikingly, these patterns of cell loss were all concentrated in the upper layers of the cortex, suggesting a cascade of effects where loss of certain particularly vulnerable cells leads to loss of their neighbors over time.
These findings not only map the progression of Alzheimer’s but also suggest potential avenues for early intervention.
“One of the challenges to diagnosing and treating Alzheimer’s is that much of the damage to the brain happens well before symptoms occur. The ability to detect these early changes means that, for the first time, we can see what is happening to a person’s brain during the earliest periods of the disease,” said Richard J. Hodes, M.D., director, NIH National Institute on Aging. “The results fundamentally alter scientists’ understanding of how Alzheimer’s harms the brain and will guide the development of new treatments for this devastating disorder.”
Can we stop Alzheimer’s first domino from falling?
The data from this study suggest a narrative for how AD unfolds, the authors said:
Changes in the brain’s immune and support cells promote, or dysregulate, inflammation. This is associated with early loss of the SST neurons, which are uniquely poised to regulate the balance of excitation and inhibition in the cortex. They also participate in attention, processing of sensory inputs, and coordinating long-distance communication across different brain regions.
The researchers hypothesize losing these cells may start a domino effect, disrupting the delicate balance between inhibition and excitation and prompting widespread system failures that ultimately lead to widespread neuronal loss and cognitive decline seen in AD.
This view of Alzheimer’s as a circuit disorder that ultimately disrupts cognition is speculative, Lein stressed. But the new study’s cellular mapping supports reframing Alzheimer’s beyond just a pathology of misfolded proteins. “The initial triggers of disease may involve pathological proteins or microglial activation, but it is loss of specific types of neurons and the connections they make that lead to cognitive decline.” This finer-grained understanding could also be crucial for developing new treatments, Lein added. Current therapies that target AD’s hallmarks—amyloid plaques and tau tangles—often show modest effectiveness with serious side effects.
If we could instead prevent the first cellular dominos from falling, perhaps we could prevent broader degeneration.
“Armed with this information, maybe we could target not only molecules like tau and amyloid, but also vulnerable cell types,” Lein said. “Perhaps we could protect them and prevent their degeneration—and the whole downstream cascade of events.”
Research reported in this publication was supported by the NIH’s National Institute on Aging (U19AG060909, P30AG066509, U19AG066567). The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
Citations
about the allen institute
Allen Institute is a 501(c)(3) nonprofit medical research organization dedicated to accelerating science for a healthier world. Through large-scale, multidisciplinary research initiatives, the Institute generates foundational knowledge, data, tools, and models that are shared openly with the world to advance our understanding of life and health. Founded by Jody Allen and the late Paul G. Allen, Allen Institute is supported primarily by the Fund for Science and Technology.

