
in this article
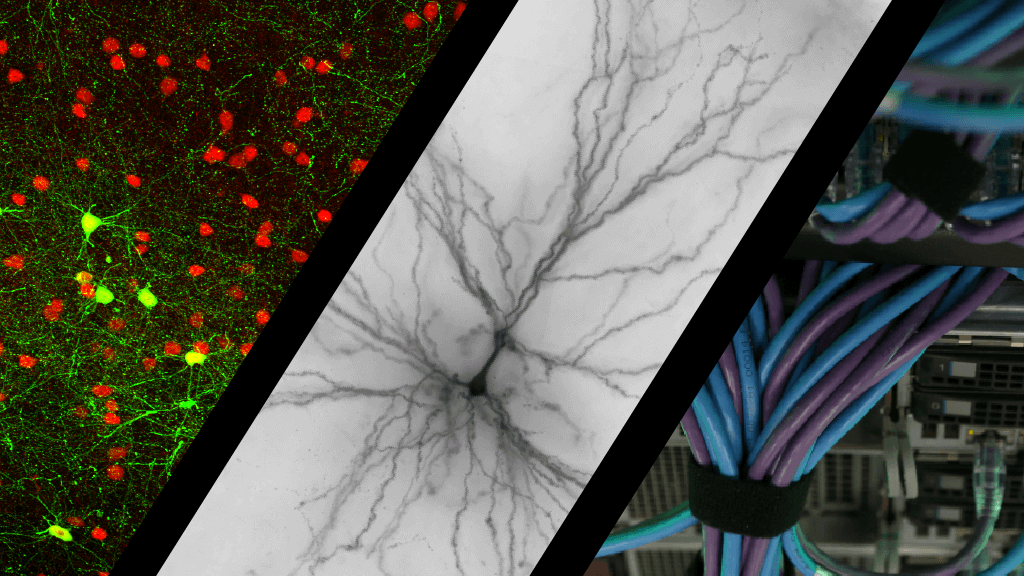
The National Institutes of Health today awarded the Allen Institute for Brain Science three five-year grants totaling nearly $100 million, to expand upon work in investigating cell types in the mouse and human brains: a crucial step toward understanding brain health and disease. The grants are part of the BRAIN Initiative Cell Census Network (BICCN), which aims to establish reference cell atlases of the brain by supporting collaborations that will create resources to be shared throughout the research community. Of the nine collaborative consortia established by these BICCN grants, three will be led by Allen Institute for Brain Science researchers.
“We are enormously honored to receive these grants, and to be the hub for these ambitious collaborative consortia that will build upon our pursuit of cataloging the cellular building blocks of the brain,” says Christof Koch, Ph.D., Chief Scientist and President of the Allen Institute for Brain Science. “Our team science and open science model enables us to create and share resources that serve the larger scientific community. Through these grants, we will be able to work closely with our collaborators to establish standards and generate resources that will propel neuroscience in exciting new directions.”
“A detailed census of the structure and function of specific cell types is one of the most promising avenues for advancing our understanding of the human brain,” says Allan Jones, Ph.D., President and CEO of the Allen Institute. “We are proud to have established ourselves in this area with our work in creating the Allen Cell Types Database, and are honored to be recognized through these grants as a leader in the field.”
BRAIN Initiative Cell Census Network Grants
“A comprehensive whole-brain atlas of cell types in the mouse” — $65.5M
The goal of this project is to create a comprehensive atlas of cell types in the whole mouse brain, the first in mammals. Researchers will conduct large-scale single-cell gene expression analysis across the entire brain along with systematic sampling of neuronal shape and connectivity in a wide range of brain areas. Researchers will analyze the correspondence of molecular, anatomical, and functional properties in the same cells, to gain an integrated understanding of cell types. Finally, a census of the number and location of cells for each type and new genetic tools targeting selected cell types will be generated.
“Building a comprehensive atlas of cell types requires a collaborative, multi-disciplinary approach,” says Hongkui Zeng, Ph.D., Executive Director of Structured Science at the Allen Institute for Brain Science. “Joining us in this endeavor are globally recognized leaders in the fields of transcriptomics, neuroanatomy, neurophysiology, computational biology and microscopy. Our collective efforts will accelerate the entire community’s progress in decoding brain function and impact everything from disease research to artificial intelligence.”
The project will be led by six Principal Investigators (PI):
- Hongkui Zeng, Project Director (PD), Allen Institute for Brain Science
- David Anderson, California Institute of Technology
- Xiaowei Zhuang, Harvard University
- Andreas Tolias, Baylor College of Medicine
- John Ngai, University of California, Berkeley
- Lior Pachter, California Institute of Technology
This large project also includes Co-Investigators from other institutions: Massachusetts Institute of Technology, Huazhong University of Science and Technology, Karolinska Institute, University of Tubingen, Weill Cornell Medicine, and collaborators from the Howard Hughes Medical Institute Janelia Research Campus.
“A multimodal atlas of human brain cell types” — $19.4M
Allen Institute researchers will lead the creation of a multimodal atlas of human brain cell types, beginning with a gene expression survey of single cells in the brain and spinal cord, and deeper analysis of samples from the cortex and hippocampus. To move from a classification to a quantitative census, the team will analyze human neurosurgical and postmortem tissue samples, and characterize the spatial distribution and relative proportions of cells expressing different sets of genes. The second part of the project will form an international consortium of experts in studying and modeling live human cells, in order to classify cells based on shape, physiology and connective properties, and then correlate these properties to gene expression data.
“This collaboration brings together world leaders in single cell transcriptomics, human cellular physiology and anatomy, and human neuron modeling for an ambitious effort to study the structure and function of the human brain itself,” says Ed Lein, Ph.D., Investigator at the Allen Institute for Brain Science. “This combined expertise and the Allen Institute’s demonstrated ability to tackle large scale research projects will enable us to build a human brain cell type atlas that will be enormously beneficial to basic and clinical research. Very little research of this sort is currently being done in the United States, and we hope to join forces to better understand both conserved and unique features of human brain relevant to human-specific diseases.”
The project will be led by Ed Lein (PI) and the following Co-Investigators at their respective Institutions:
- Sten Linnarsson, Karolinska Institute
- Gábor Tamás, University of Szeged
- Huib Mansvelder, VU University Amsterdam
- Idan Segev, The Hebrew University of Jerusalem
“A community resource for single cell data in the brain” — $14.5M
In collaboration with external partners, the Allen Institute will develop and house the BRAIN Cell Data Center (BCDC). This foundational community resource will include a web-portal for sharing single cell brain data, tools and knowledge generated by BRAIN Initiative Cell Census Network partners. The BCDC will also develop and deploy fundamental data models, common community standards and scientific results.
“The BRAIN Cell Data Center will be an incredibly valuable resource for the entire neuroscience community and beyond,” says Michael Hawrylycz, Ph.D., Investigator at the Allen Institute for Brain Science. “Applying cell type semantic and spatial community standards to these data, and making them available online, provides a tremendous opportunity for improving our understanding of the diverse cell types in the brain and how they’re organized.”
The project will be led by five Principal Investigators (PI):
- Michael Hawrylycz, Allen Institute for Brain Science
- Lydia Ng, Allen Institute for Brain Science
- Anthony Philippakis, Broad Institute of MIT and Harvard
- Maryann Martone, University of California, San Diego
- James Gee, University of Pennsylvania
About the Allen Institute for Brain Science
The Allen Institute for Brain Science is a division of the Allen Institute (alleninstitute.org), an independent, 501(c)(3) nonprofit medical research organization, and is dedicated to accelerating the understanding of how the human brain works in health and disease. Using a big science approach, the Allen Institute generates useful public resources used by researchers and organizations around the globe, drives technological and analytical advances, and discovers fundamental brain properties through integration of experiments, modeling and theory. Launched in 2003 with a seed contribution from founder and philanthropist Paul G. Allen, the Allen Institute is supported by a diversity of government, foundation and private funds to enable its projects. Given the Institute’s achievements, Mr. Allen committed an additional $300 million in 2012 for the first four years of a ten-year plan to further propel and expand the Institute’s scientific programs, bringing his total commitment to date to $500 million. The Allen Institute for Brain Science’s data and tools are publicly available online at brain-map.org.
Citations
about the allen institute
The Allen Institute is an independent, 501(c)(3) nonprofit research organization founded by philanthropist and visionary, the late Paul G. Allen. The Allen Institute is dedicated to answering some of the biggest questions in bioscience and accelerating research worldwide. The Institute is a recognized leader in large-scale research with a commitment to an open science model. For more information, visit alleninstitute.org.