in this article
authors
Stem cells are the adaptable all-stars of the human body. Adult stem cells help maintain and repair certain tissues such as skin and intestines. Human-induced pluripotent stem cells, or iPSCs, are different; they’re made by reprogramming a patient’s own cells into cells that can become nearly any cell type. Because of that shapeshifting property, they hold tremendous promise for regenerative medicine and the repair of damaged tissues and organs. They’re also key players in cancer and Alzheimer’s research, among other diseases.

Having such a versatile biological tool is critical for research. But before scientists can use iPSCs, they need to grow and multiply them into larger numbers, and during that process many of these cells acquire genetic defects. One of the most common is trisomy 12, where a cell has three copies of chromosome 12 instead of the usual two. Trisomy 12 is associated with multiple types of cancer, making it a major concern not only because it can distort experimental results, but also because it may compromise the safety and usefulness of iPSCs in new therapies.
what scientists discovered about trisomy 12
Researchers thought they knew how trisomy 12 developed, but findings published in the Journal of Cell Biology reshaped that understanding. Previously, scientists believed the genetic defect was the result of a rare mistake in a single cell that gradually spread because cells carrying the extra chromosome grew faster than normal.
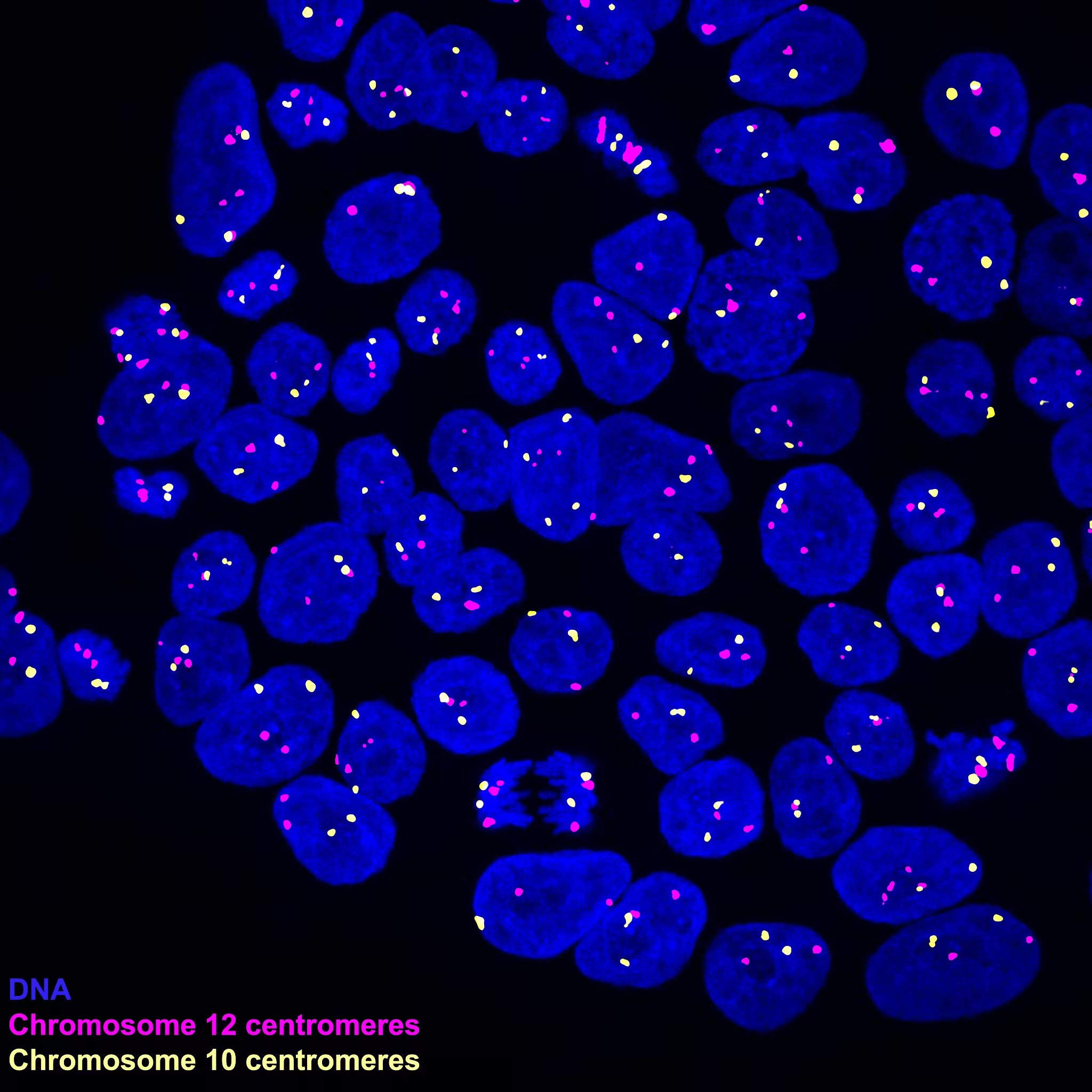
But researchers at the Oklahoma Medical Research Foundation (OMRF) discovered that trisomy 12 appears simultaneously in many cells and spreads quickly during the various phases of growth. “In our study, we saw trisomy 12 presence increase from about five percent of cells to almost 100 percent in a period of 40 days,” said Maria Narozna, a researcher at OMRF and co-author of the study.
Narozna and her team discovered that chromosome 12 — one of the tiny packages of DNA inside our cells — is especially prone to mistakes, known as missegregation, when cells divide. Normally, when a cell divides into two it first makes a full copy of every chromosome and then splits into two cells so that each cell has one copy of the complete chromosome. But where there is a trisomy 12 defect, chromosome 12 gets "stuck," like a rope pulled between two sides in a tug of war. This results in two chromosomes that are tied together with the second copy unable to migrate to the new cell, leaving it trapped in a tiny DNA structure called the micronucleus. This leaves one of the cells with an extra, incomplete piece of chromosome 12. When that cell divides again, the chromosome 12 trapped in the micronucleus is added back into the new cell, giving it three copies of chromosome 12 instead of the normal two.
Scientists calculated that about one to two out of every 100 normal cells made this genetic mistake each time they divided, which in the world of cell biology is a very high number. “Given how many cell divisions occur in the human body, a one- to two-percent conversion rate for every passage would be catastrophic to a person,” said Gary Gorbsky, OMRF professor and study co-author.
The study findings can help scientists better understand and perhaps treat cancer. Trisomy 12 cells are frequently found in chronic lymphocytic leukemia (CLL) and other cancers, suggesting that these study findings may help researchers better understand chromosome abnormalities beyond stem cell cultures. Normally, if a cell has an extra chromosome, the body detects that something is wrong with it and tells that cell to stop dividing, like an automatic shutoff mechanism. But cells with the trisomy 12 abnormality have figured out a way to ignore those orders and keep growing and may even gain a growth advantage. By better understanding these "rule-breakers," scientists may eventually find ways to detect, limit, or prevent them from occurring.
Researchers also studied why chromosome 12 is more likely to develop abnormalities. Chromosomes have a protective cap at the end of each strand known as a telomere. Think of it like the plastic at the end of a shoelace keeping it from fraying. Telomeres protect chromosomes from degradation, but chromosome 12 has one of the shortest “protective caps” in the human genome, making it susceptible to damage when cells are duplicating their DNA before division.
how researchers used Allen Institute tools and resources
Researchers from the OMRF used Allen Institute’s cell lines AICS 0012 and AICS 0013 in their study. AICS 0012 proved especially valuable because it could, in some situations and specific conditions, reproduce the trisomy 12 defect. This allowed scientists to track that process with high precision and uncover the origin and mechanisms behind the defect. AICS 0013, which reveals the nuclear protein of the cell, helped researchers determine whether the tiny DNA structures (micronuclei), where chromosome 12 often became trapped, were stable or unstable.
“We’re very grateful to the Allen Institute for developing these cell lines because they gave us a powerful human stem cell system in which we could follow key mitotic and nuclear structures in living cells,” said Gorbsky. “More importantly, these models are far better than cancer cell lines for studying how cells acquire the wrong number of chromosomes. In cancer cells, chromosome abnormalities are often so numerous that it becomes very difficult to trace where the problem began or which change is driving the biology.”
Citations
about the allen institute
The Allen Institute is an independent, 501(c)(3) nonprofit research organization founded by philanthropist and visionary, the late Paul G. Allen. The Allen Institute is dedicated to answering some of the biggest questions in bioscience and accelerating research worldwide. The Institute is a recognized leader in large-scale research with a commitment to an open science model. For more information, visit alleninstitute.org.